Two Gene Mutations Linked with Telomeres in Pulmonary Fibrosis
Written by |
Hard at work in the laboratory of Dr. Christine Kim Garcia, Associate Professor of Internal Medicine at UT Southwestern Medical Center, is a group of researchers studying familial pulmonary fibrosis. The disease is characterized by fatal lung scarring and is now associated with mutations in two genes: PARN and RTEL1. Knowing more about these mutations will allow researchers to develop cures for familial pulmonary fibrosis.
“Our ultimate goal is to gain a full understanding of what causes the genetic form of this disease so that effective medications can be developed,” said Dr. Garcia in a news release from UT Southwestern.
To begin achieving this goal, Dr. Garcia’s laboratory conducted a study entitled “Exome Sequencing Links Mutations in PARN and RTEL1 with Familial Pulmonary Fibrosis and Telomere Shortening,” published in Nature Genetics. Using exome sequencing of nearly 100 individuals who identified a case of pulmonary fibrosis in the family, the team found that approximately 7% of familial pulmonary fibrosis is caused by mutations in PARN and RTEL1.
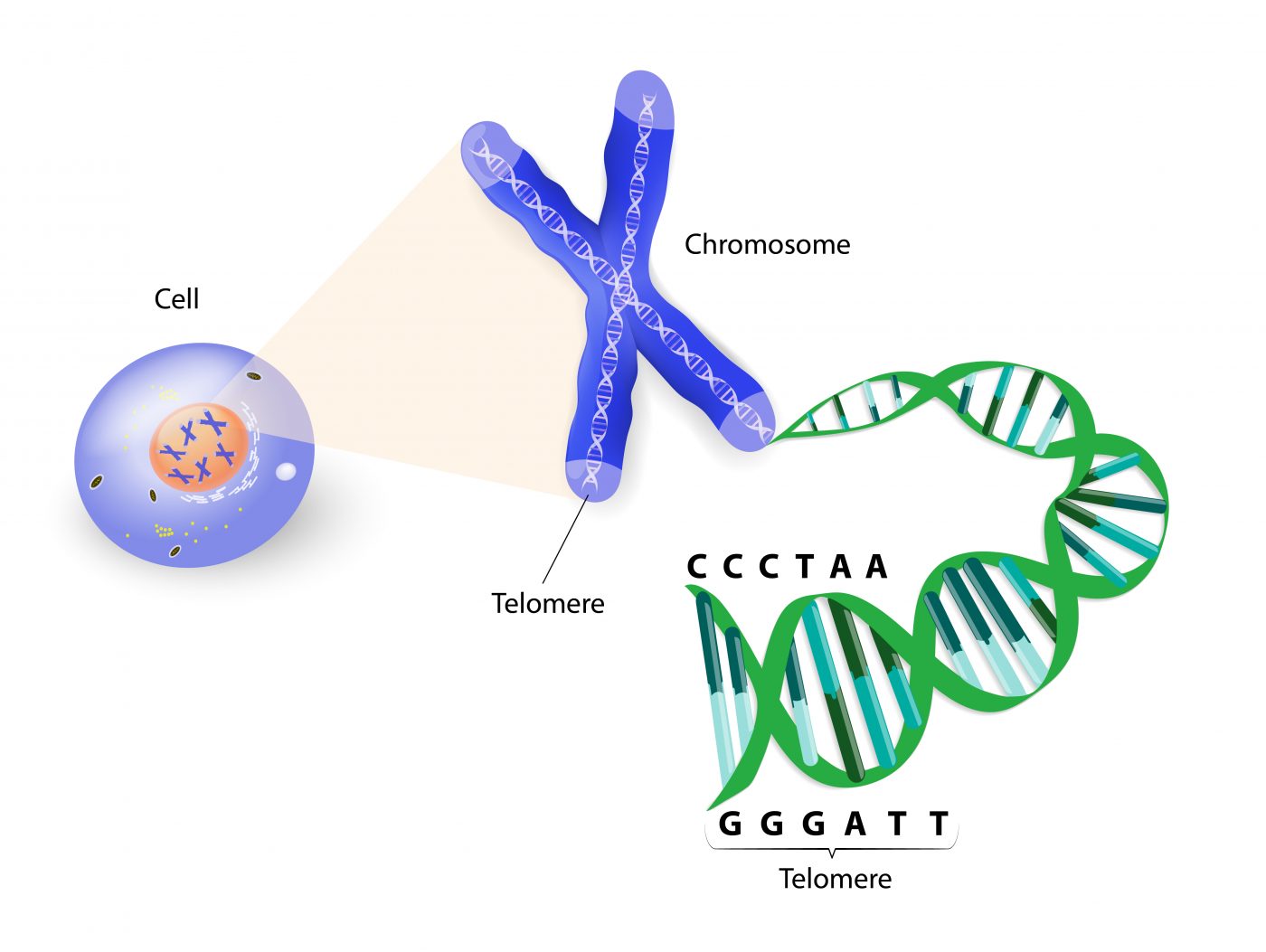
The team then sought to draw an association between the genes and the lengths of patients’ telomeres, which are the genetic caps on the ends of chromosomes to prevent DNA damage. They used real-time polymerase chain reaction (quantitative PCR) to measure telomere length. Patients who carried the PARN and RTEL1 mutations had shortened telomeres. Their families were also more likely to have epigenetic changes in telomere length.
“Although RTEL1 had been previously linked to telomere biology, our finding that PARN was involved in telomere regulation and human disease was completely unexpected,” commented Dr. Garcia. “There were statistically more mutations found in these two genes than you would expect by chance.”
“We found that the mean, age-adjusted telomere length of all rare variant carriers was significantly shorter than normal controls,” continued Dr. Bridget Stuart, lead author on the paper and an Assistant Professor of Pediatrics. “This finding implicates both genes in telomere maintenance as well as development of pulmonary fibrosis.”
Finding cures based on these mutations may alleviate some of the disease burden on the 50,000 Americans living with idiopathic pulmonary fibrosis, of which 1 in 20 have familial pulmonary fibrosis. Currently, unless patients receive a lung transplant, they typically die within three years of being diagnosed with pulmonary fibrosis.
Glossary of Terms:
[wikibox lang=”en”]telomere[/wikibox]
[wikibox lang=”en”]Poly(A)-specific ribonuclease[/wikibox]



