Gene Therapy Targeting Cause of Cellular Aging Treats Pulmonary Fibrosis in Mouse Study
Written by |
Looking at pulmonary fibrosis (PF) as an age-related disease and working with a gene therapy that lengthens the life of lung cells, researchers report that they were able to lessen — and even stop — lung fibrosis in mice.
“This is the first time that pulmonary fibrosis has been treated as an age-related disease, looking for rejuvenating the affected tissues,” Maria A. Blasco, study’s lead author and group leader at the Spanish National Cancer Research Centre (CNIO), in Madrid, said in a press release.
The study “Therapeutic effects of telomerase in mice with pulmonary fibrosis induced by damage to the lungs and short telomeres” was published in the journal eLife.
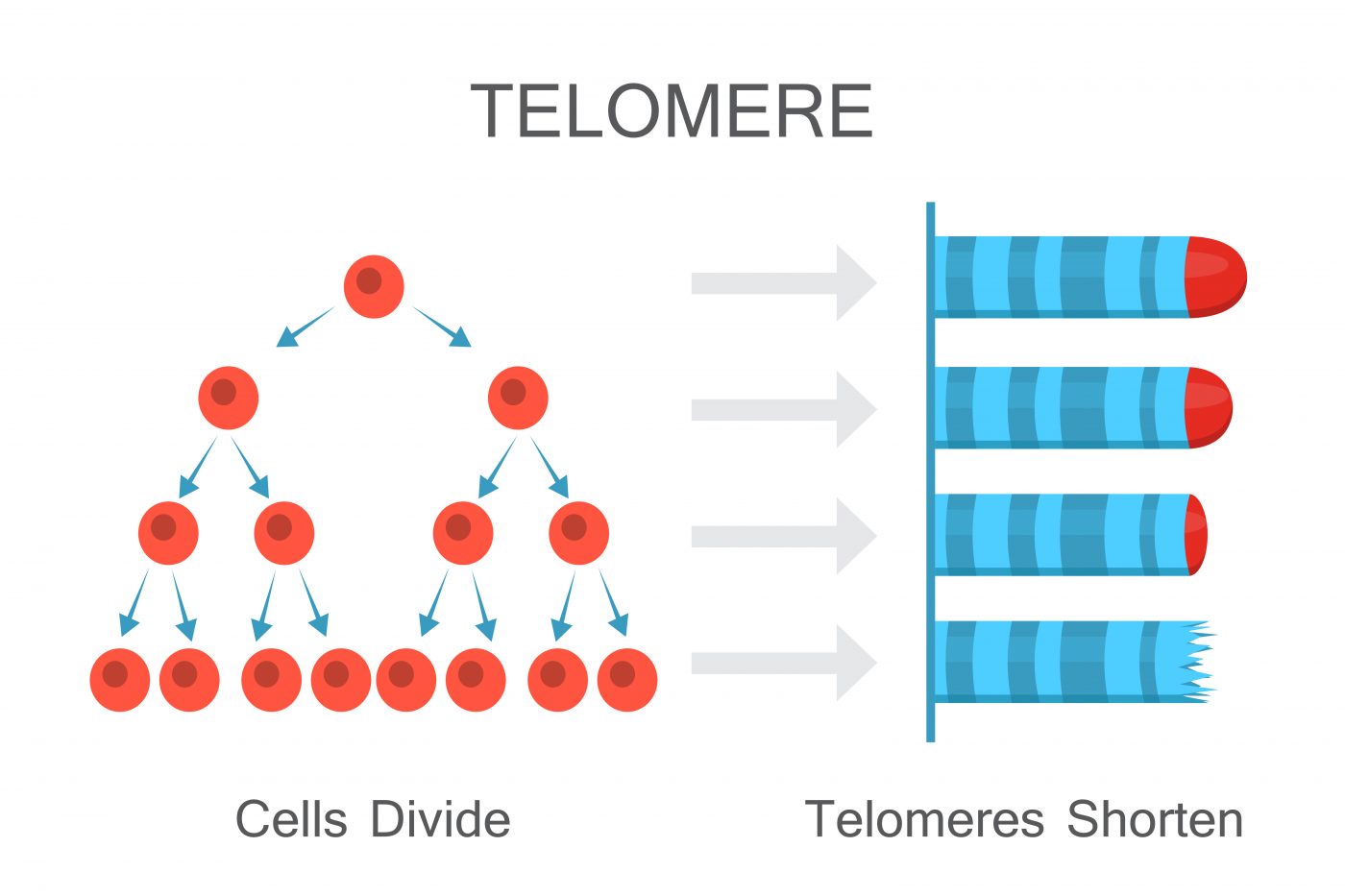
Telomeres are the protein structures located at the end of the tip of chromosomes, long seen as the clock that counts cells’ division and lifespan. They work as caps that protect the chromosomes from damage, much like the aglets in the shoelaces. But as time goes by, they become shorter and are no longer able to fulfill this protective role.
As a result, chromosomes accumulate damage and cells stop dividing. This hampers tissue regeneration, and is one of the mechanisms triggering aging.
Several diseases have been linked with this telomeric shortening and PF is one of them. Patients with hereditary and non-hereditary PF carry shorter telomeres, and the cells response to counteract this shortening may actually promote the disease. Cells have the capacity to counter telomere’s shortening but in PF this response may actually activate a “repair program”d that culminates in tissue scaring.
The research team at CNIO Telomere and Telomerase Group, led my Blasco, developed a new mouse model for PF that reproduces in full the human PF disease — including tissue scaring and telomere shortening. Currently, the most-used model is the bleomycin-induced PF model, with bleomycin inducing damage in the lungs. But in this mouse PF model, remission is witnessed within several weeks of disease induction and there is no telomere shortening.
The study describing the new mouse model was published in Cell Reports in 2015, titled “Mice with Pulmonary Fibrosis Driven by Telomere Dysfunction.”
“This is an essential model to test therapeutic strategies based on the activation of telomerase [the enzyme that repairs the telomeres],” the researchers wrote at that time.
In their new study, they show that activating the enzyme telomerase to increase the length of telomeres improved lung function and lessened inflammation and fibrosis in this new mouse model. After eight weeks of treatment, fibrosis continued to improve and even disappear.
The treatment was based on gene therapy where researchers used a virus (AAV9 viral vector), modified to cause no harm to humans, to introduce the telomerase gene into the lung cells. Mice received one injection of the gene therapy.
“We observe that telomerase gene therapy reverses the fibrotic process in mice, which suggests that it could be effective in human patients, opening a new therapeutic opportunity towards the treatment of this disease,” said Juan Manuel Povedano, the study’s co-first author.
The study was carried in close collaboration with gene therapy expert Fàtima Bosch with the Autonomous University of Barcelona. Bosch is already considering the potential clinical applications of these findings.
“The strategy devised by the CNIO group is very encouraging,” Bosch said. “Although we are still far from reaching the clinic, we are already generating gene therapy vectors for human therapy.”
“The most relevant aspect of our work is that it suggests a potentially viable and effective solution to a real clinical problem, pulmonary fibrosis, for which there is still no treatment,” said Paula Martínez, co-first author of the study. “The only approved treatments for pulmonary fibrosis up-to-date have no curative effects, as they target a symptom and not the cause of fibrosis.”



