Underlying Lung Fibrosis Mechanisms Linked to Mitochondria Dysfunction
Written by |
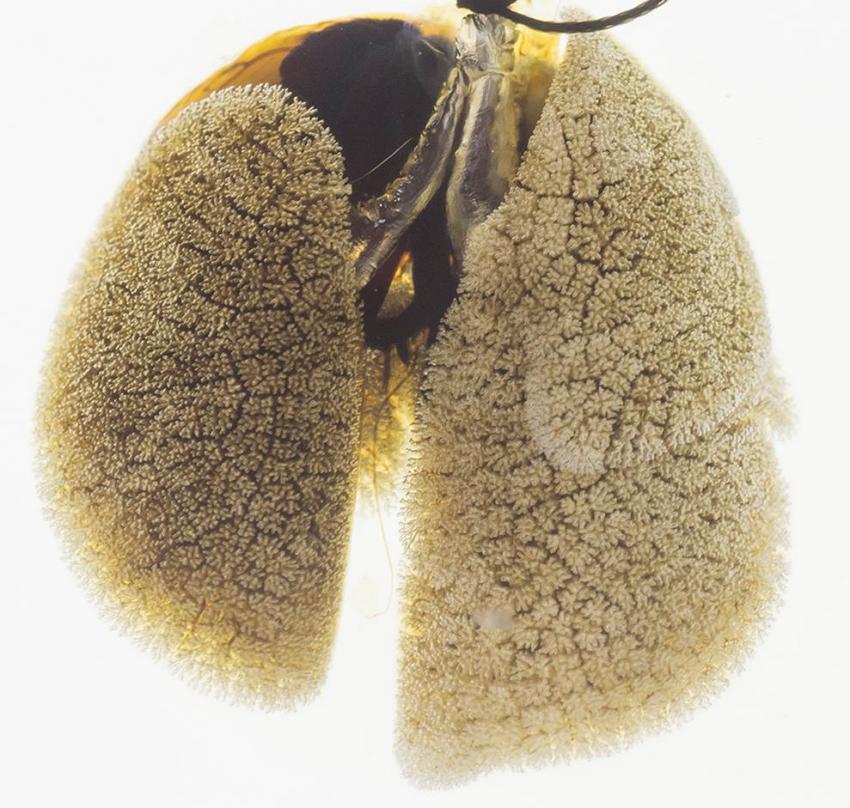
Pattern of alveolar oxygen exchange units on the surface of a mouse lung. Credit: Dr. Shou Ping Guan
 A new study entitled “PINK1 deficiency impairs mitochondrial homeostasis and promotes lung fibrosis” discovered mitochondria PINK1 impaired expression underlies the formation of fibrotic tissue in older patients lungs. The study was published in The Journal of Clinical Investigation.
A new study entitled “PINK1 deficiency impairs mitochondrial homeostasis and promotes lung fibrosis” discovered mitochondria PINK1 impaired expression underlies the formation of fibrotic tissue in older patients lungs. The study was published in The Journal of Clinical Investigation.
Idiopathic pulmonary fibrosis (IPF) is a chronic disease characterized by the formation of scars in lung tissue (a process termed as fibrosis) leading to a progressive decline in lung function. The disease usually affects older adults (people 75 years old or older have a significantly higher incidence, 50 times higher, than younger adults), where it ultimately becomes fatal; however, the mechanisms underlying the effect of age on lung-decreased function are largely unknown.
In this new study, a team of researchers at the University of Pittsburgh School of Medicine hypothesized that a mechanism triggered by aging and observed in other diseases, neurodegenerative and cardiovascular, could also impact lung fibrosis. The mechanism is a decline in mitochondrial function, the key organelle responsible for providing energy for the cell. The authors found that indeed mitochondrial decline in lung cells – the alveolar type II cells (AECIIs) – promote lung fibrosis in aging lungs. They observed an accumulation of dysmorphic and dysfunctional mitochondria in the lungs of IPF patients, which were also found in mice with advance age. The decline in mitochondria occurred in association with increased expression of stress markers in another cellular organelle, the Endoplasmic Reticulum, a condition termed as ER stress. The team performed further studies and determined the levels of a mitochondrial enzyme, the PTEN-induced putative kinase 1 (PINK1), whose functions include maintaining mitochondrial morphology and function. Aging lungs exhibited lower levels of PINK1. Knocking down PINK1 expression in lung epithelial cells, as well as mice lacking PINK1, showed mitochondria dysfunction and a higher susceptibility to develop lung fibrosis. Accordingly, the expression of profibrotic factors, such as TGF-β, was found increased in alveolar cells with reduced PINK1 expression.
[adrotate group=”3″]
Therefore, the authors discovered that aging lungs exhibit PINK1 expression impairment resulting in mitochondrial dysfunction and apoptosis, a process that culminates in the formation of fibrotic tissue in lungs.
Ana L. Mora, M.D., assistant professor in the Division of Pulmonary, Allergy and Critical Care Medicine and a member of the Heart, Lung, Blood and Vascular Medicine Institute (VMI) at Pitt commented in a press release “We found that low PINK1 is associated with increasing age and cellular stress. This might help explain why older people are at greater risk for developing IPF, and it could mean developing drugs that can boost PINK1 levels or improve mitochondrial function will help treat IPF.”
Mark Gladwin, M.D., professor and chief, Division of Pulmonary, Allergy and Critical Care Medicine, and VMI director added, “These findings are remarkable as they identify a similar disease pathway to that seen in other age related brain diseases. This is the first study to find that the mitochondria themselves, the energy factories of our cells, are altered with lung fibrosis.”



